The second ionization energy tends to increase across a period and decrease down a group however, ionization involving core electrons are higher energy than those involving valence electrons. The second ionization energy is the energy required to the remove the second of two electrons from an atom. The availability of a second valence electron gives Mg (Group 2) the lowest second ionization energy. Table 20.3.1 Selected Properties of the Group 1 Elements. As a result, Cs (melting point 28.5C) is one of only three metals (the others are Ga and Hg) that are liquids at body temperature (37C). 
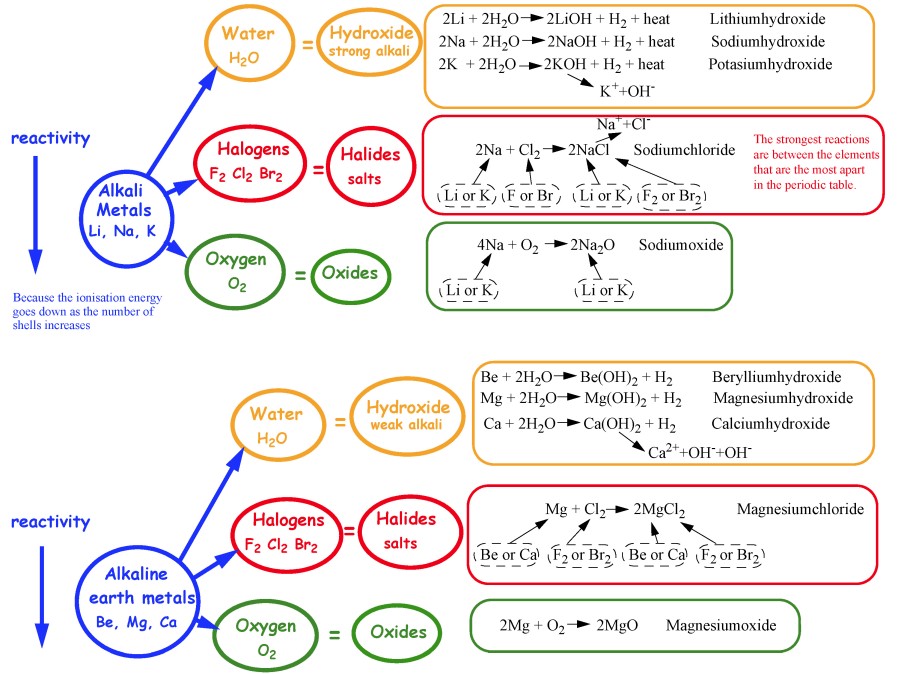
However, the second ionization energy of Na (Group 1) is much higher than that of Mg (Group 2) because Na has only 1 valence electron and removing a second electron from Na requires the loss of a core electron. An unusual trend in the group 1 elements is the smooth decrease in the melting and boiling points from Li to Cs. If comparing elements in a period (row) three, the nonmetals Si (Group 14) and S (Group 16) are farther to the right of the period and both have a higher second ionization energy than the metals Na and Mg. select all the correct statements concerning valence electrons of elements. add the additional electron configuration needed to reach the element on the periodic table. write the symbol of the noble gas in brackets. find the closest noble gas in the period above the element. Beryllium reacts with steam at high temperatures (typically around 700☌ or more) to give white beryllium oxide and. The alkali and alkaline earth metals comprise the first two columns of the periodic table, traditionally referred to as Groups IA and IIA, respectively (Figure 1). locate the position of the element on the periodic table. It uses these reactions to explore the trend in reactivity in Group 2. 
Beryllium (Be) is the only alkaline earth metal that, even when heated to red heat, does not react with water or steam. This page looks at the reactions of the Group 2 elements - beryllium, magnesium, calcium, strontium and barium - with water (or steam). The same is true for 2nd ionization energy except when the second electron being removed is NOT a valence electron. Similar to Group 1 metals, alkaline earth metals (Be, Mg, Ca, Sr, Ba, and Ra) are the second most reactive metals in the periodic table, increasing reactivity as the period increases. Group 2 First ionization energies tend to increase across a row and decrease down a column.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
